Medical MIM parts are small, complex, precision metal components used in endoscopic instruments, dental systems, surgical tools, diagnostic equipment, and compact medical mechanisms. This page is a medical parts overview, not a deep process article. It helps engineers and sourcing teams see which medical part categories may fit metal injection molding, which categories deserve dedicated subpages, and what information should be prepared before a drawing review. MIM is usually considered when a part is small, geometrically complex, difficult to machine efficiently, and produced in repeatable volume. The key decision is not whether the part is “medical,” but whether its geometry, material, tolerance, surface condition, annual volume, and inspection requirements fit the MIM route.
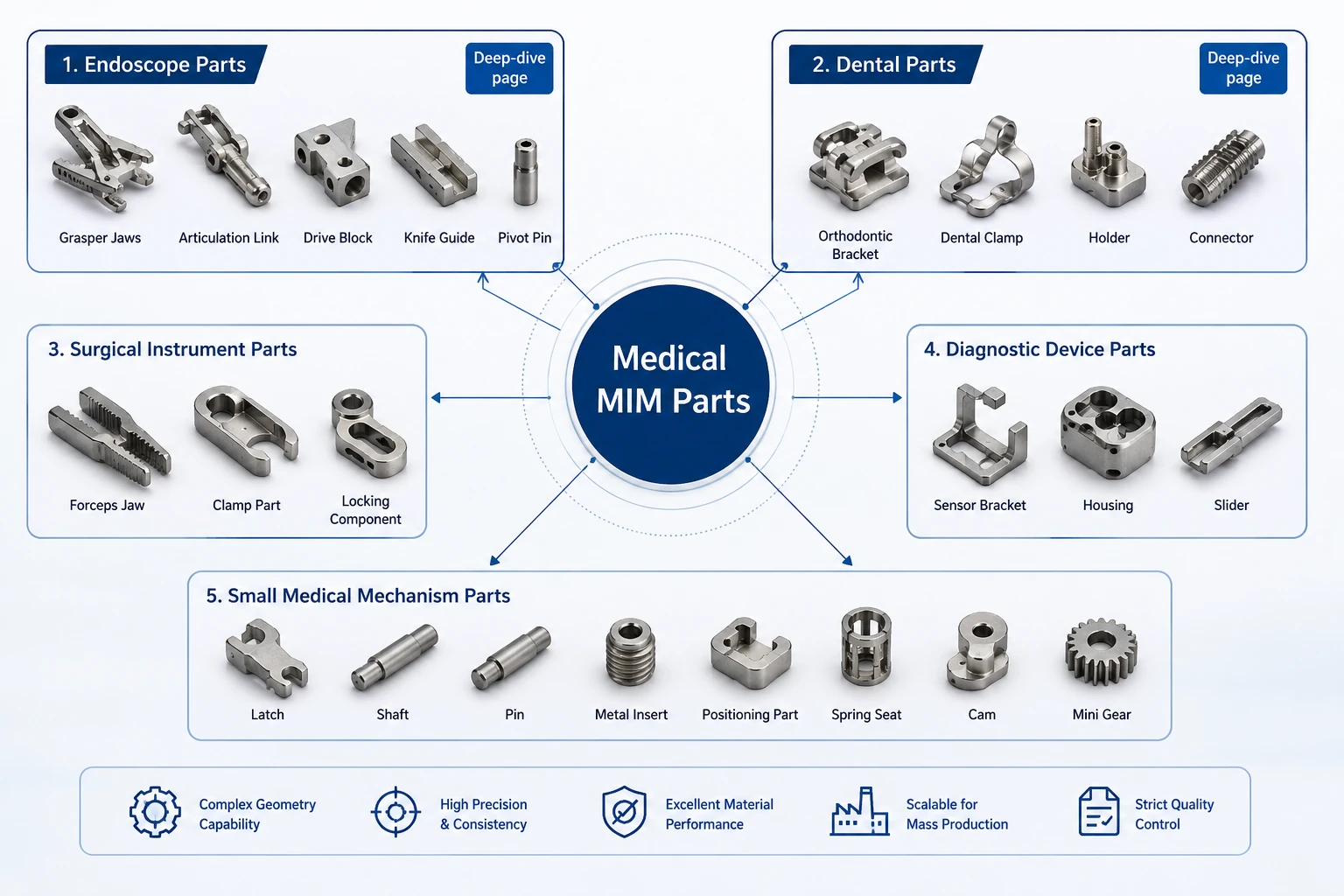
Medical MIM parts should be grouped by device use and engineering function, not by a single product name. This overview introduces the main medical part categories and guides users to the right deeper page or RFQ path when more detailed review is needed.
Medical MIM Parts
Medical MIM parts are typically small metal components that combine fine features, complex geometry, and production repeatability. The MIM route uses fine metal powder mixed with binder into feedstock, followed by injection molding, green part handling, debinding, and sintering. Because the part shrinks during sintering, tooling compensation and dimensional control must be reviewed before production.
For a medical parts overview page, the main job is to show part categories and direct users to the right next step. Detailed process explanation belongs on the metal injection molding process page. Detailed endoscope and dental part discussion belongs on the dedicated pages.
This page helps users identify whether their medical component belongs to Endoscope Parts, Dental Parts, surgical instrument parts, diagnostic device parts, or small medical mechanism parts before they submit a drawing.
Common Medical MIM Part Categories
Medical MIM parts should be organized by part category and engineering requirement. Not every category needs a separate page at the beginning. Endoscope Parts and Dental Parts have clearer independent search intent and enough DFM depth to support dedicated pages. Other medical part categories can be displayed on this overview page until search data, project cases, or image resources justify deeper pages.

Only part families with clear search intent and enough engineering depth should become dedicated subpages. This structure prevents thin pages and keeps the Medical Parts page focused on category navigation, suitability screening, and RFQ preparation.
| Medical Part Category | Typical MIM Parts | Why MIM May Fit | Recommended Next Step |
|---|---|---|---|
| Endoscope Parts | Grasper jaws, biopsy forceps parts, articulation links, drive blocks, knife guides, pivot pins | Small size, fine features, holes, slots, compact motion structures | View dedicated page |
| Dental Parts | Orthodontic brackets, self-ligating parts, dental tool parts, holders, clamps, connectors | Small geometry, repeatability, surface finish, corrosion resistance | View dedicated page |
| Surgical Instrument Parts | Forceps components, clamp parts, scissors components, locking parts, small handles | Strength, repeatable shape, compact functional surfaces | Covered on this overview page first |
| Diagnostic Device Parts | Holders, small housings, sliders, sensor brackets, linkages | Compact mechanisms, small metal features, stable production | Covered on this overview page |
| Drug Delivery and Small Medical Mechanism Parts | Latch parts, release mechanisms, metal inserts, positioning parts | Small movement parts and compact metal structures | Covered carefully on this overview page |
| Orthopedic Tool Parts | Guide parts, clamps, compact tool inserts, locking components | Strong small metal parts for instruments and fixtures | Covered without implant qualification claims |
Endoscope Parts
Endoscope parts are one of the strongest deeper-page directions under Medical Parts. Typical components include grasper jaws, biopsy forceps components, articulation links, drive blocks, knife guides, and pivot pins. These parts often include small holes, thin sections, guide slots, mating surfaces, and burr-sensitive edges.
On this overview page, the goal is only to introduce the category and guide users to the deeper page. Detailed discussion of jaw alignment, linkage distortion, slot accuracy, cutting edge condition, and sintering support should be handled on the Endoscope MIM Parts page.

Endoscope parts should be reviewed by geometry, movement, mating surfaces, and critical dimensions. This image explains why Endoscope Parts deserves a deeper page while this Medical Parts page remains a category-level overview.
Recommended next step: visit Endoscope Parts if your project includes jaws, links, drive blocks, knife guides, or miniature motion components.
Dental Parts
Dental Parts is the second priority deeper-page direction under Medical Parts. Typical MIM candidates include orthodontic brackets, self-ligating bracket parts, dental instrument components, small holders, clamps, and compact connectors. These parts are usually small, detailed, and sensitive to surface finish, edge condition, and repeatability.
This overview page should not go deeply into bracket slot design, base design, polishing strategy, or self-ligating structures. Those topics belong on the Dental MIM Parts page. Implant-related components should only be reviewed through project-specific material, validation, and regulatory requirements.

Dental MIM parts need functional-zone review before tooling. This image supports the routing logic: Dental Parts has enough structural and surface-review depth to deserve a dedicated page, while this page keeps the category introduction concise.
Other Medical Device Part Categories
The following medical part categories are important to show, but they do not need separate pages at the current stage. Keeping them on this overview page avoids thin content and keeps the medical parts structure clean.
Forceps components, clamp parts, scissors components, locking parts, jaw or grip parts, small handle inserts, and trigger elements may fit MIM when the geometry is compact and production volume supports tooling.
Review focus: edge condition, gripping features, mating surfaces, burr control, and functional wear areas.
Small housings, holders, sensor brackets, sliders, compact connectors, and linkages should be reviewed by geometry, mating surfaces, and inspection requirements rather than device name alone.
Review focus: sensor location, assembly fit, thin walls, small bosses, holder stability, and inspection method.
Latch parts, release elements, small locking components, inserts, shafts, and positioning parts may fit MIM when they are small metal mechanism components. Complete device-level manufacturing or drug-contact qualification requires separate project review.
Review focus: movement path, locking force, sliding contact, mating part condition, and component-level material requirements.
Guide parts, clamps, compact tool inserts, and locking elements may be considered when they are non-implant tool or fixture components. Implantable use requires separate project validation.
Review focus: load direction, guide surface, clamp engagement, edge control, and post-processing requirements.
Medical assemblies may include small rotating, locating, locking, or pivoting parts. For cross-industry part-family discussion, see MIM shafts and pins.
Review focus: diameter tolerance, straightness, pivot fit, wear surface, and secondary machining needs.
If the main concern is tolerance, critical dimensions, or inspection method, review high precision MIM parts instead of forcing all details into this overview page.
Review focus: critical dimensions, tolerance stack-up, inspection datum, functional surfaces, and post-sintering correction.
Materials and Surface Review
Medical MIM parts often use stainless steels or other customer-specified alloys, but material choice should follow the drawing, application environment, mechanical requirement, cleaning exposure, and surface requirement. This page only introduces the material logic. Detailed grade discussion belongs on the stainless steel MIM materials page.
316L stainless steel is often reviewed when corrosion resistance and cleaning exposure are important. 17-4PH stainless steel may be reviewed when higher strength is required. Final material selection must follow the customer drawing and project conditions.
Medical parts may require deburring, polishing, passivation, edge control, surface roughness definition, and separation of cosmetic surfaces from functional surfaces. For finishing-related review, see surface finishing capability.
If corrosion exposure is a primary requirement, the project can also be reviewed through corrosion-resistant MIM parts. If sliding, gripping, locking, or pivoting surfaces are critical, see wear-resistant MIM parts.
When Medical Parts Fit MIM
MIM should be selected by part geometry, production volume, material requirement, and tolerance strategy. A medical part is not automatically a MIM candidate just because it is small or used in a medical device.

MIM suitability depends on geometry, production volume, and functional requirements. This matrix helps users filter projects before RFQ and supports fast engineering screening before deeper review or quotation.
Medical MIM Suitability Filter
Before requesting a quote, engineers can use the following filter to judge whether a medical component is likely to deserve MIM review.
| Filter Item | What to Review | Positive Signal for MIM Review |
|---|---|---|
| Part size | Overall size, wall thickness, section balance, and part weight. | Compact metal part with features that are difficult to machine one by one. |
| Feature complexity | Slots, holes, teeth, ribs, undercuts, guide surfaces, and compact motion features. | Several small features can be formed near-net-shape instead of separate CNC operations. |
| Annual volume | Expected production quantity, validation stage, and long-term demand. | Production volume is high enough to justify tooling and process development. |
| Tolerance strategy | Critical dimensions, inspection datum, mating surfaces, and tolerance stack-up. | Only selected functional areas require tight control, while general geometry can use MIM tolerance planning. |
| Secondary operation dependency | Machining, polishing, passivation, grinding, threading, or special inspection needs. | Secondary operations are limited to critical areas instead of nearly every surface. |
| Good Fit for MIM | Poor Fit for MIM |
|---|---|
| Small metal part with complex features such as slots, holes, ribs, teeth, or compact motion geometry. | Large simple geometry that can be machined, stamped, cast, or fabricated more economically. |
| Medium to high annual volume where tooling investment can be justified. | Very low-volume prototype or design still changing frequently. |
| Part requires repeatable metal strength, corrosion resistance, or functional surfaces. | Almost every surface requires tight CNC finishing, removing the MIM cost advantage. |
| Several features can be formed near-net-shape instead of machined one by one. | Implantable or regulated use without project-specific material, validation, and quality documentation. |
In practice, MIM is a near-net-shape process. Critical holes, threads, bearing areas, sliding faces, sealing faces, or sharp functional edges may still require secondary machining, grinding, polishing, passivation, or inspection. For tolerance planning, review the MIM tolerance and shrinkage checklist.
Medical MIM Part Review Checklist
A useful medical MIM review should start with the drawing, not only a part name. The purpose of this checklist is to help engineers and sourcing teams prepare enough information for a realistic MIM feasibility review.

A medical MIM quotation is only reliable when drawing, material, tolerances, surface requirements, and production volume are reviewed together. This checklist helps users prepare complete RFQ information before contacting XTMIM.
| Review Input | Why It Matters |
|---|---|
| 2D drawing and 3D CAD file | Needed to evaluate geometry, tooling direction, shrinkage compensation, and critical features. |
| Material requirement | Material affects sintering behavior, strength, corrosion resistance, finishing, and inspection strategy. |
| Critical dimensions and tolerances | Helps separate functional dimensions from general dimensions before tooling review. |
| Surface finish, edge, and burr requirements | Important for small medical mechanisms, mating surfaces, cleaning exposure, and assembly function. |
| Annual volume and project stage | Determines whether MIM tooling and development cost are suitable for the project. |
| Secondary operations and inspection expectations | Needed to confirm machining, polishing, passivation, inspection, and documentation requirements. |
Useful preparation paths include submit drawing for review, the RFQ preparation guide, and request a quote.
Request a Medical MIM Part Review
If your project includes a small complex metal part for an endoscope device, dental system, surgical instrument, diagnostic device, or compact medical mechanism, the next step is a drawing-based review. XTMIM can review whether the part geometry, material, tolerance strategy, surface finish, secondary operations, and inspection expectations are suitable for MIM.
For a useful review, provide the 2D drawing, 3D CAD file, expected material, annual volume, critical dimensions, surface finish requirements, edge or burr requirements, and application background. This helps identify tooling, sintering, finishing, and inspection risks before production planning.
XTMIM reviews and manufactures precision MIM components. Device-level regulatory approval, sterilization validation, clinical performance, and final product responsibility remain project-specific and should be confirmed by the device owner or qualified medical device manufacturer.
FAQ
What medical parts can be made by MIM?
MIM can be used for small, complex metal parts in endoscopic devices, dental systems, surgical instruments, diagnostic equipment, drug delivery mechanisms, and compact medical assemblies. Typical parts include jaws, links, drive blocks, brackets, holders, clamps, locking parts, shafts, pins, and small connectors. Final suitability depends on geometry, material, tolerance, surface finish, annual volume, and inspection requirements.
Is MIM suitable for endoscope parts?
MIM can be suitable for endoscope parts such as grasper jaws, biopsy forceps components, articulation links, drive blocks, knife guides, and pivot features. These parts often include small holes, slots, thin sections, and compact mechanisms. However, edge condition, burr control, hole position, and mating surfaces should be reviewed before tooling.
Can dental parts be made by metal injection molding?
Yes, some dental and orthodontic parts can be made by MIM, especially small precision components such as orthodontic brackets, self-ligating elements, dental tool parts, holders, clamps, and connectors. Implantable dental components should be reviewed separately according to customer specifications, material documentation, validation expectations, and applicable regulatory requirements.
Are all medical parts suitable for MIM?
No. MIM is not suitable for every medical part. Large simple parts, very low-volume parts, frequently changing designs, and components where nearly every surface requires tight CNC finishing may not be good fits. MIM is strongest for small, complex, repeatable metal parts where near-net-shape forming reduces machining burden.
What information is needed for a medical MIM part review?
A useful review usually requires a 2D drawing, 3D CAD file, material requirement, expected annual volume, critical dimensions, surface finish requirements, edge or burr requirements, secondary machining needs, and application background. Without these inputs, only a general feasibility opinion can be provided.
Does XTMIM manufacture complete medical devices?
No. This page focuses on precision MIM component manufacturability and component-level production review. Complete medical device design, device-level regulatory approval, sterilization validation, clinical performance, and final product responsibility should be managed by the device owner or qualified medical device manufacturer.
Reviewed by XTMIM Engineering Team
This content is prepared and reviewed from the perspective of MIM process suitability, medical part geometry, material selection, DFM review, tooling risk, sintering shrinkage, tolerance planning, surface finishing, inspection requirements, and production feasibility.
The review focuses on whether the part geometry is suitable for MIM, which features may create tooling or sintering risk, whether material selection matches the part function, which tolerances require special review, whether secondary machining or polishing may be needed, and what information should be provided before RFQ.
Standards and Technical Reference Note
Medical MIM part evaluation should combine material standards, MIM process knowledge, customer drawings, and project-specific DFM review. Standards can guide material and quality discussions, but they do not replace supplier-specific process review, customer validation, or device-level regulatory responsibility.
XTMIM’s role on this page is component-level MIM manufacturability review. Medical device regulatory approval, sterilization validation, and final product responsibility remain project-specific and should be confirmed by the device owner or qualified medical device manufacturer.
- MPIF Standard 35-MIM: relevant for material specification and engineering property discussion for metal injection molded parts.
- MIMA Endoscopic Device Parts case reference: useful for understanding why endoscopic device components can be treated as a deeper medical MIM part family.
- EPMA MIM process reference: useful for understanding MIM as a process for complex metal parts and for distinguishing MIM from conventional pressed-and-sintered powder metallurgy.
- ISO 13485 context: relevant to medical device quality management expectations, but should not be used as a supplier certification claim unless verified for the specific company and project.
